Buy Fentanyl Precursor 4-ANPP online
Buy Fentanyl Precursor 4-ANPP online
4-ANPP, also known as 4-anilino-N-phenethylpiperidine (4-ANPP), 4-aminophenyl-1-phenethylpiperidine, or despropionyl fentanyl,[3] is a direct precursor to fentanyl and acetylfentanyl. It is commonly found as a contaminant in samples of drugs containing fentanyl, which may include samples represented by the supplier as heroin or other opioids.[4] It is not psychoactive and is present only as a result of improper chemical purification.
4-ANPP is useful in the synthesis of pharmaceuticals, primarily fentanyl and related analogs. Paul Janssen (founder of Janssen Pharmaceutica) first synthesized fentanyl in 1960 using a similar method, with Benzylfentanyl as an intermediate.[5] The following synthesis, developed by an individual under the pseudonym of Siegfried, involves the reductive amination of N–phenethyl-4-piperidinone (NPP) with aniline to make to 4-ANPP. This product is reacted with propionyl chloride or acetyl chloride to form either fentanyl or acetylfentanyl.
21409-26-7
-
-
Basic information
- Product Name: 4-AMINOPHENYL-1-PHENETHYLPIPERIDINE
- Synonyms: N-PHENYL-1-(2-PHENYLETHYL) PIPERIDIN-4-AMINE;(1-PHENETHYL-PIPERIDIN-4-YL)-PHENYL-AMINE;N-Phenyl-1-(2-phenethyl)piperidin-4-amine;N-(1-PHENETHYL-PIPERIDIN-4-YL)-ANILINE;N-[1-(2-Phenylethyl)-4-piperidinyl]benzenamine;N-Phenyl-1-(2-phenylethyl)-4-piperidinamine;4-Piperidinamine, N-phenyl-1-(2-phenylethyl)-;N-Phenyl-N’-[1-(2-phenylethyl)]-4-piperidine Discontinued See: A625875 Buy Fentanyl Precursor 4-ANPP online
- CAS NO:21409-26-7
- Molecular Formula: C19H24N2
- Molecular Weight: 280.40726
- EINECS: N/A
- Product Categories: Aromatics;Metabolite & Impurities;Heterocycles;Intermediates & Fine Chemicals;Pharmaceuticals
- Mol File: 21409-26-7.mol
-
Chemical Properties
- Melting Point: 94-96°C
- Boiling Point: 424.604 °C at 760 mmHg
- Flash Point: 161.923 °C
- Appearance: Pale yellow solid
- Density: 1.075 g/cm3
- Vapor Pressure: 2.04E-07mmHg at 25°C
- Refractive Index: 1.603
- Storage Temp.: Hygroscopic, -20°C Freezer, Under Inert Atmosphere
- Solubility: N/A
- PKA: 9.03±0.10(Predicted)
- CAS DataBase Reference: 4-AMINOPHENYL-1-PHENETHYLPIPERIDINE(CAS DataBase Reference)
- NIST Chemistry Reference: 4-AMINOPHENYL-1-PHENETHYLPIPERIDINE(21409-26-7)
- EPA Substance Registry System: 4-AMINOPHENYL-1-PHENETHYLPIPERIDINE(21409-26-7)
-
Safety Data
- Hazard Codes: N/A
- Statements: 22-36/37/38
- Safety Statements: 22-37/39-46
- RIDADR: UN1230 – class 3 – PG 2 – Methanol, solution
- WGK Germany: 2
- RTECS:
- HazardClass: N/A
- PackingGroup: N/A
- Hazardous Substances Data: 21409-26-7(Hazardous Substances Data)
21409-26-7 Suppliers
 This product is a nationally controlled contraband or patented product, and the Lookchem platform doesn’t provide relevant sales information.
This product is a nationally controlled contraband or patented product, and the Lookchem platform doesn’t provide relevant sales information.21409-26-7 Usage
Uses
Used in Pharmaceutical Synthesis:
4-AMINOPHENYL-1-PHENETHYLPIPERIDINE is used as a chemical intermediate for the synthesis of fentanyl and related opioids. It plays a crucial role in the production of potent analgesic medications used in medical settings for pain management.
Used in Research Applications:
4-AMINOPHENYL-1-PHENETHYLPIPERIDINE is used as an analytical reference material for research purposes. It aids scientists in understanding the chemical properties and metabolic pathways of opioids, contributing to the development of new drugs and therapies.
Used in Forensic Applications:
4-AMINOPHENYL-1-PHENETHYLPIPERIDINE is utilized as a certified reference material in forensic analysis. It helps in the identification and quantification of opioids in criminal investigations, particularly in cases involving illicit drug trade and substance abuse.
Used in Metabolite Studies:
4-AMINOPHENYL-1-PHENETHYLPIPERIDINE is used as a metabolite of acetyl fentanyl, butyryl fentanyl, furanyl fentanyl, acrylfentanyl, and fentanyl. Studying its presence and behavior in biological systems can provide insights into the metabolism and potential toxic effects of these opioids. Buy Fentanyl Precursor 4-ANPP online
Check Digit Verification of cas no
The CAS Registry Mumber 21409-26-7 includes 8 digits separated into 3 groups by hyphens. The first part of the number,starting from the left, has 5 digits, 2,1,4,0 and 9 respectively; the second part has 2 digits, 2 and 6 respectively.
Calculate Digit Verification of CAS Registry Number 21409-26:
(7*2)+(6*1)+(5*4)+(4*0)+(3*9)+(2*2)+(1*6)=77
77 % 10 = 7
So 21409-26-7 is a valid CAS Registry Number.
21409-26-7SDS
SAFETY DATA SHEETS
According to Globally Harmonized System of Classification and Labelling of Chemicals (GHS) – Sixth revised edition
Version: 1.0
Creation Date: Aug 15, 2017
Revision Date: Aug 15, 2017
1.Identification
1.1 GHS Product identifier
| Product name | 4-Aminophenyl-1-phenethylpiperidine |
|---|
1.2 Other means of identification
| Product number | – |
|---|---|
| Other names | N-phenyl-1-(2-phenylethyl)piperidin-4-amine |
1.3 Recommended use of the chemical and restrictions on use
| Identified uses | For industry use only. |
|---|---|
| Uses advised against | no data available |
1.4 Supplier’s details
| Buy Fentanyl Precursor 4-ANPP online | |
|---|---|
| Buy Fentanyl Precursor 4-ANPP online | |
| Buy Fentanyl Precursor 4-ANPP online | |
| Buy Fentanyl Precursor 4-ANPP online |
1.5 Emergency phone number
| Emergency phone number | –Buy Fentanyl Precursor 4-ANPP online |
|---|---|
| Service hours | Monday to Friday, 9am-5pm (Standard time zone: UTC/GMT +8 hours). |
More Details:21409-26-7 SDS
21409-26-7Upstream product
21409-26-7Downstream Products
- 82003-76-73-Oxo-N-(1-phenethyl-piperidin-4-yl)-N-phenyl-butyramide
- 82003-73-42-chloro-N-phenyl-N-<1-(2-phenylethyl)-4-piperidinyl>acetamide
- 437-38-7N-phenyl-N-[1-(2-phenylethyl)-4-piperidinyl]propanamide
- 912286-42-13-(1,3-dioxo-1,3-dihydro-isoindol-2-yl)-N-(1-phenethyl-piperidin-4-yl)-N-phenyl-propionamide
- 52994-23-7Carboxyfentanyl
- 87640-69-54-(2-chloroethyl)-1-(2-phenylethyl)-3-phenyltetrahydropyrimidin-2(1H)-one
- 122861-38-5N-<1-(2-phenylethyl)-4-piperidyl>-N-phenylthiopropanamide
- 1184873-14-0N-[1-(2-phenylethyl)-piperidin-4-yl]-nitrosoaniline
- 1338727-47-1tryptophan 3,5-bis(trifluoromethyl)benzyl ester 4-oxo-4-((1-phenethylpiperidin-4-yl)(phenyl)amino)butanoate
- 1338727-48-2tryptophan 3,5-bis(trifluoromethyl)benzyl ester 5-oxo-5-((1-phenethylpiperidin-4-yl)(phenyl)amino)pentanoate
- 1338727-49-3tryptophan 3,5-bis(trifluoromethyl)benzyl ester 2-(2-oxo-2-((1-phenethylpiperidin-4-yl)(phenyl)amino)ethoxy)acetate
- 1338727-32-4C23H27N2O2(1+)*ClHO4*ClO4(1-)
- 1338727-35-7C24H29N2O2(1+)*ClHO4*ClO4(1-)
- 1338727-38-0C23H27N2O3(1+)*ClHO4*ClO4(1-)
21409-26-7 Relevant academic research and scientific papers
Evaluation of agonistic activity of fluorinated and nonfluorinated fentanyl analogs on μ-opioid receptor using a cell-based assay system
The agonistic activity of fluorinated and nonfluorinated fentanyl analogs on μ-opioid receptor was investigated using a cell-based assay system. Based on the activity, fentanyl analogs were ranked as follows: fentanyl>isobutyrylfentanyl≈butyrylfentanyl≈methoxyacetylfentanyl>acetylfentanyl. However, among the fentanyl analogs fluorinated on the Nphenyl ring, 2-fluoro analogs and 3-fluoro analogs showed the strongest and weakest activities, respectively. These results suggest that the 2-fluorinated isomers of fentanyl analogs are more likely to cause poisoning.
ADJUVANTED CONJUGATE OPIOID VACCINE
The adjuvanted conjugate opioid vaccine described herein is a conjugate of a protein carrier and at least one opioid backbone component or hapten conjugated thereto, admixed with at least one adjuvant. Anti-opioid effects are demonstrated after administration of a vaccine made up of the CRM197 protein carrier linked to a FEN backbone, combined with adjuvants such as dmLT or LTA1.
Chemoselective Reductive Aminations in Aqueous Nanoreactors Using Parts per Million Level Pd/C Catalysis
Casotti, Gianluca,Gao, Eugene S.,Jin, Henry S.,Lipshutz, Bruce H.,Takale, Balaram S.,Thakore, Ruchita R.
supporting information,
Condensation in recyclable water between aldehydes or ketones and amines occurs smoothly within the hydrophobic cores of nanomicelles, resulting in imine formation that is subject to subsequent reduction leading, overall, to reductive amination. This micellar technology enables the synthesis of several types of pharmaceuticals, a new procedure that relies on only 2000 ppm (0.20 mol %) palladium from commercially available Pd/C. A broad range of substrates can be used under mild conditions, leading to high chemical yields of the desired secondary and tertiary amines.
Fentanyl family at the mu-opioid receptor: Uniform assessment of binding and computational analysis
Interactions of 21 fentanyl derivatives with μ-opioid receptor (μOR) were studied using experimental and theoretical methods. Their binding to μOR was assessed with radioligand competitive binding assay. A uniform set of binding affinity data contains val
Metabolism of butyrylfentanyl in fresh human hepatocytes: Chemical synthesis of authentic metabolite standards for definitive identification
The metabolism of butyrylfentanyl, a new designer drug, was investigated using fresh human hepatocytes isolated from a liver-humanized mouse model. In the culture medium of hepatocytes incubated with butyrylfentanyl, the desphenethylated metabolite (nor-butyrylfentanyl), w-hydroxy-butyrylfentanyl, (w-1)-hydroxy-butyrylfentanyl, 4′-hydroxy-butyrylfentanyl, β-hydroxy-butyrylfentanyl, 4′-hydroxy-3′- methoxy-butyrylfentanyl, and w-carboxy-fentanyl were identified as the metabolites of butyrylfentanyl. Each metabolite was definitively identified by comparing the analytical data with those of authentic standards. The amount of the main metabolite, nor-butyrylfentanyl, reached 37% of the initial amount of butyrylfentanyl at 48 h. W-Hydroxy-butyrylfentanyl and (w-1)-hydroxy-butyrylfentanyl, formed by hydroxylation at the Nbutyryl group of butyrylfentanyl, were the second and third largest metabolites, respectively. The majority of 4′-hydroxy-butyrylfentanyl and 4′-hydroxy-3′-methoxy-butyrylfentanyl was considered to be conjugated. CYP reaction phenotyping for butyrylfentanyl using human liver microsomes and various anti-CYP antibodies revealed that CYP3A4 was involved in the formation of nor-butyrylfentanyl, (w-1)-hydroxybutyrylfentanyl, and β-hydroxy-butyrylfentanyl. In contrast, CYP2D6 was involved in the formation of w-hydroxy-butyrylfentanyl.
Fentanyl, also known as ‘jackpot’, is a synthetic opiate that is 50–100 times more potent than morphine. Clandestine laboratories produce analogues of fentanyl, known as fentalogues to circumvent legislation regarding its production. Three pyridyl fentalogues were synthesized and then hyperpolarized by signal amplification by reversible exchange (SABRE) to appraise the forensic potential of the technique. A maximum enhancement of -168-fold at 1.4 T was recorded for the ortho pyridyl 1H nuclei. Studies of the activation parameters for the three fentalogues revealed that the ratio of ligand loss trans to hydride and hydride loss in the complex [Ir(IMes)(L)3(H)2]+ (IMes=1,3-bis(2,4,6-trimethylphenyl)imidazole-2-ylidene) ranged from 0.52 to 1.83. The fentalogue possessing the ratio closest to unity produced the largest enhancement subsequent to performing SABRE at earth’s magnetic field. It was possible to hyperpolarize a pyridyl fentalogue selectively from a matrix that consisted largely of heroin (97 : 3 heroin:fentalogue) to validate the use of SABRE as a forensic tool.
A 4 – anilino – N – phenethyl piperidine simple synthesis of process
The invention discloses a 4 – anilino – N – phenethyl piperidine simple synthesis process, comprising the following steps: the reaction bottle, adding raw material N – phenethyl – 4 – piperidone and organic solvent dichloromethane, opening stirring, after
Metabolism of fentanyl and acetylfentanyl in human-induced pluripotent stem cell-derived hepatocytes
To evaluate the capability of human-induced pluripotent stem cell-derived hepatocytes (h-iPS-HEP) in drug metabolism, the profiles of the metabolites of fentanyl, a powerful synthetic opioid, and acetylfentanyl, an N-acetyl analog of fentanyl, in the cells were determined and analyzed. Commercially available h-iPSHEP were incubated with fentanyl or acetylfentanyl for 24 or 48 h. After enzymatic hydrolysis, the medium was deproteinized with acetonitrile, then analyzed by LC/MS. Desphenethylated metabolites and some hydroxylated metabolites, including 4′-hydroxy-fentanyl and β-hydroxy-fentanyl, were detected as metabolites of fentanyl and acetylfentanyl in the medium. The main metabolite of fentanyl with h-iPS-HEP was the desphenethylated metabolite, which was in agreement with in vivo results. These results suggest that h-iPSHEP may be useful as a tool for investigating drug metabolism.
N-phenethyl-4-aniline-based process for preparation of piperidines
–
The invention relates to a method for preparing N-phenethyl-4-phenylaminopiperidine. The method comprises the main steps of carrying out hydrogenation and amination on N-phenethyl-4-piperidone (structural formula as shown in a formula (II)) and aniline (structural formula as shown in a formula (III)) in ethanol in the presence of Raney Ni at the temperature of 50-100 DEG C so as to obtain the target product. The method for preparing N-phenethyl-4-phenylaminopiperidine has the advantages of cheap and available raw material, less byproduct, good product purity, high yield, low cost and the like.
Palladium-catalyzed hydroaminocarbonylation of alkenes with amines promoted by weak acid
The weak acid has been identified as an efficient basicity-mask to overcome the basicity barrier imparted by aliphatic amines in the Pd-catalyzed hydroaminocarbonylation, which enables both aromatic and aliphatic amines to be applicable in the palladium-catalyzed hydroaminocarbonylation reaction. Notably, by using this protocol, the marketed herbicide of Propanil and drug of Fentanyl could be easily obtained in a one-pot manner. Buy Fentanyl Precursor 4-ANPP online
Order 4-Anilino-N-phenethylpiperidine (4-ANPP) Precursor
Order 4-Anilino-N-phenethylpiperidine (4-ANPP) Precursor
Buy desproprionyl fentanyl, 4-anilino-N-phenethylpiperidine, 4-ANPP, ANPP,
4-ANPP, also known as 4-anilino-N-phenethylpiperidine (4-ANPP), 4-aminophenyl-1-phenethylpiperidine, or despropionyl fentanyl,[3] is a direct precursor to fentanyl and acetylfentanyl. It is commonly found as a contaminant in samples of drugs containing fentanyl, which may include samples represented by the supplier as heroin or other opioids.[4] It is not psychoactive and is present only as a result of improper chemical purification. Order 4-Anilino-N-phenethylpiperidine (4-ANPP) Precursor
4-ANPP is useful in the synthesis of pharmaceuticals, primarily fentanyl and related analogs. Paul Janssen (founder of Janssen Pharmaceutica) first synthesized fentanyl in 1960 using a similar method, with Benzylfentanyl as an intermediate.[5] The following synthesis, developed by an individual under the pseudonym of Siegfried, involves the reductive amination of N–phenethyl-4-piperidinone (NPP) with aniline to make to 4-ANPP. This product is reacted with propionyl chloride or acetyl chloride to form either fentanyl or acetylfentanyl. Order 4-Anilino-N-phenethylpiperidine (4-ANPP) Precursor
Buy 4-Anilino-N-phenethylpiperidine (4-ANPP)
Buy 4-Anilino-N-phenethylpiperidine (4-ANPP). 4-ANPP-13C6 (CRM) is a certified reference material intended for use as an internal standard for the quantification of 4-ANPP by GC- or LC-MS. 4-ANPP is categorized as a piperidinamine. It is an intermediate in the synthesis of fentanyl from N-phenethyl-4-piperidone .1 As such, 4-ANPP has been used as a precursor for the manufacture of fentanyl and related opioids. 4-ANPP is also an impurity found in fentanyl preparations. It is a known metabolite of acetyl fentanyl , butyryl fentanyl , furanyl fentanyl , acrylfentanyl , and fentanyl.2,3,4,5 4-ANPP-13C6 is regulated as a Schedule II compound in the United States. This product is intended for research and forensic applications. Buy 4-Anilino-N-phenethylpiperidine (4-ANPP)
4-ANPP, also known as 4-anilino-N-phenethylpiperidine (4-ANPP), 4-aminophenyl-1-phenethylpiperidine, or despropionyl fentanyl,[3] is a direct precursor to fentanyl and acetylfentanyl. It is commonly found as a contaminant in samples of drugs containing fentanyl, which may include samples represented by the supplier as heroin or other opioids.[4] It is not psychoactive and is present only as a result of improper chemical purification. Buy 4-Anilino-N-phenethylpiperidine (4-ANPP)
4-ANPP is useful in the synthesis of pharmaceuticals, primarily fentanyl and related analogs. Paul Janssen (founder of Janssen Pharmaceutica) first synthesized fentanyl in 1960 using a similar method, with Benzylfentanyl as an intermediate.[5] The following synthesis, developed by an individual under the pseudonym of Siegfried, involves the reductive amination of N–phenethyl-4-piperidinone (NPP) with aniline to make to 4-ANPP. This product is reacted with propionyl chloride or acetyl chloride to form either fentanyl or acetylfentanyl. Order 4-Anilino-N-phenethylpiperidine (4-ANPP) Precursor
Buy desproprionyl fentanyl, 4-anilino-N-phenethylpiperidine, 4-ANPP, ANPP
Buy desproprionyl fentanyl, 4-anilino-N-phenethylpiperidine, 4-ANPP, ANPP
Buy desproprionyl fentanyl, 4-anilino-N-phenethylpiperidine, 4-ANPP, ANPP
Buy desproprionyl fentanyl, 4-anilino-N-phenethylpiperidine, 4-ANPP, ANPP
Buy desproprionyl fentanyl, 4-anilino-N-phenethylpiperidine, 4-ANPP, ANPP
Buy desproprionyl fentanyl, 4-anilino-N-phenethylpiperidine, 4-ANPP, ANPP
Buy desproprionyl fentanyl, 4-anilino-N-phenethylpiperidine, 4-ANPP, ANPP
Buy desproprionyl fentanyl, 4-anilino-N-phenethylpiperidine, 4-ANPP, ANPP
Buy desproprionyl fentanyl, 4-anilino-N-phenethylpiperidine, 4-ANPP, ANPP
Buy desproprionyl fentanyl, 4-anilino-N-phenethylpiperidine, 4-ANPP, ANPP
Buy desproprionyl fentanyl, 4-anilino-N-phenethylpiperidine, 4-ANPP, ANPP
Buy desproprionyl fentanyl, 4-anilino-N-phenethylpiperidine, 4-ANPP, ANPP
- 4-Aminophenyl-1-phenethylpiperidine-13C6
- 4-Anilino-N-phenethylpiperidine-13C6
- Despropionyl fentanyl-13C6
Technical Information
Regulatory Information
Shipping & Storage Information
Buy 4-Anilino-N-phenethylpiperidine (4-ANPP)
Buy 4-Anilino-N-phenethylpiperidine (4-ANPP)
Buy 4-Anilino-N-phenethylpiperidine (4-ANPP)
Buy 4-Anilino-N-phenethylpiperidine (4-ANPP)
Buy 4-Anilino-N-phenethylpiperidine (4-ANPP)
Buy 4-Anilino-N-phenethylpiperidine (4-ANPP)
Buy 4-Anilino-N-phenethylpiperidine (4-ANPP)
Buy 4-Anilino-N-phenethylpiperidine (4-ANPP)
Buy 4-Anilino-N-phenethylpiperidine (4-ANPP)
Buy 4-Anilino-N-phenethylpiperidine (4-ANPP)
Buy 4-Anilino-N-phenethylpiperidine (4-ANPP)
Buy 4-Anilino-N-phenethylpiperidine (4-ANPP)
Buy 4-Anilino-N-phenethylpiperidine (4-ANPP)
Buy 4-Anilino-N-phenethylpiperidine (4-ANPP)
Buy 4-Anilino-N-phenethylpiperidine (4-ANPP)
Buy 4-Anilino-N-phenethylpiperidine (4-ANPP)
1.Pease, J.P., LePine, A.J., and Smith, C.M.Methods for preparing fentanyl and fentanyl intermediatesPatent Application PublicationUS 2013/0281702 A1(2013) 2.Watanabe, S., Vikingsson, S., Roman, M., et al.In vitro and in vivo metabolite identification studies for the new synthetic opioids acetylfentanyl, acrylfentanyl, furanylfentanyl, and 4-fluoro-isobutyrylfentanylAAPS J.19(4)1102-1122(2017) 3.Labroo, R.B., Paine, M.F., Thummel, K.E., et al.Fentanyl metabolism by human hepatic and intestinal cytochrome P450 3A4: Implications for interindividual variability in disposition, efficacy, and drug interactionsDrug Metab. Dispos.25(9)1072-1079(1997) 4.Melent’ev, A.B., Kataev, S.S., and Dvorskaya, O.N.Identification and analytical properties of acetyl fentanyl metabolitesJ. Anal. Chem.70(2)216-224(2015) 5.Steuer, A.E., Williner, E., Staeheli, S.N., et al.Studies on the metabolism of the fentanyl-derived designer drug butyrfentanyl in human in vitro liver preparations and authentic human samples using liquid chromatography-high resolution mass spectrometry (LC-HRMS)Drug Test Anal.9(7)1085-1092(2017)
4-ANPP (Item No. 18810) is an analytical reference material categorized as an opioid metabolite and a precursor in the synthesis of fentanyl (Item Nos. ISO60197 | 22659 | 14719) and other opioids. 1, 2, 3, 4, 5 4-ANPP is a metabolite of acetyl fentanyl (Item Nos. ISO60128 | ISO00128), butyryl fentanyl (Item Nos. 19734 | 14728), furanyl fentanyl (Item Nos. 19633 | 18705), acrylfentanyl (Item Nos. 23060 | 19312), and fentanyl. 1, 2, 3, 4 It has also been found as an impurity in illicit fentanyl preparations. 6 4-ANPP is regulated as a Schedule II compound in the United States.
4-ANPP (N-phenyl-1- (2-phenylethyl) -4-piperidinamine), a new opioid drug, classified as piperidinamine, synthesized as an intermediate between fentanyl from N-phenethyl-4-piperidone. 4-ANPP is a metabolite of acetylfentanil, butyrylfentanyl, furanylfentanil, acrylfentanyl and fentanyl. 4-ANPP is produced on modern chemical equipment, by professionals who know their business, which makes it clean and of high quality.
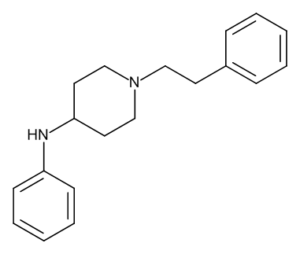
| Synonyms |
|
| IUPAC | N-phenyl-1-(2-phenylethyl)-4-piperidinamine |
| Formula | C19H24N2 |
| Molecular weight | 280.4 g/mol |
| CAS | 21409-26-7 |
| Appearance | Powder |
| Purity | ≥ 98% |
4-ANPP and other chemicals sold on this website are designed and must be used strictly for research and forensic medical examination.
Adverse properties of have not been studied.
Storage conditions of this chemical: in a cool and dry place. The stability of this chemical compound can last up to 2 years, under the right storage conditions.







Reviews
There are no reviews yet.